Learn tips about Class IV laser therapy and other health related topics on the Companion Therapy Lasers blog! Check back weekly for updated posts.
Welcome back to the Companion Animal Health Regenerative Medicine blog series! In case you missed Part 1, you can read it here. In today’s blog, we are going to answer two more of the most frequently asked questions surrounding Platelet Rich Plasma.
Question 1: What is a joint flare? How often are they seen post injection?
A joint flare is an inflammatory response in the joint due to a change in the synovial environment. Also referred to as Reactive Synovitis, joint flares can be seen after the injection or withdrawal of fluid into or from the joint. The most common characteristics are: swelling of the injected joint(s), heat emittance from the area and mild to moderate lameness. Typically lasting between 24-28 hours post injection, joint flares can be managed utilizing pain medication (excluding NSAIDs).
Joint flares are reported to occur between 15-20% of patients, but have not been reported to have a negative effect on the treatment outcome. If symptoms are lasting more than 72 hours, synovial fluid analysis is recommended in case there is an unlikely event of an immune moderated disease or septic joint.
Question 2: How long should I wait to laser the area after injecting Platelet Rich Plasma?
 Currently there is limited data surrounding the interaction of photobiomodulation and PRP. However, preliminary research is beginning to show promise for the immediate use of laser post-injection. In one recent study, researchers investigated the use of photobiomodulation, platelet rich plasma and the combination of both in the healing of an Achilles tendon. The results of this study showed compounding therapeutic effects, shortening the healing time and returning the tissue closer to “normal” tissue structure.
Currently there is limited data surrounding the interaction of photobiomodulation and PRP. However, preliminary research is beginning to show promise for the immediate use of laser post-injection. In one recent study, researchers investigated the use of photobiomodulation, platelet rich plasma and the combination of both in the healing of an Achilles tendon. The results of this study showed compounding therapeutic effects, shortening the healing time and returning the tissue closer to “normal” tissue structure.
While this study shows promise, further research and validation in canine patients will provide more accurate recommendations for post injection settings and protocols. Currently it is recommended to wait 3-4 days post injection to begin treating the area with photobiomodulation.
If you would like to learn more about Platelet Rich Plasma and Stem Cell therapies or if you have questions that were not answered in this blog, please contact Companion Animal Health at info@companiontherapies.com.
Stay tuned for our next blog which will explore the benefits of treating with Platelet Rich Plasma!
Currently, professional human athletes are benefiting from the use of laser (photobiomodulation) therapy delivery platforms. In fact, more and more professional sport franchises and Olympic teams are utilizing this modality to not only treat performance-related injuries, but to enable improved performance while minimizing potential tissue damage.
 There is a growing number of performance athletes in the canine world, in multiple disciplines such as agility, fly ball ,and Shutzhund, to name a few popular ones. These canine athletes deserve the same amount of respect and care as their human counterparts. A performance athlete will by definition face a greater risk of event-related tissue injury due to excessive or extended demand of use.
There is a growing number of performance athletes in the canine world, in multiple disciplines such as agility, fly ball ,and Shutzhund, to name a few popular ones. These canine athletes deserve the same amount of respect and care as their human counterparts. A performance athlete will by definition face a greater risk of event-related tissue injury due to excessive or extended demand of use.
The therapy laser will be beneficial in several stages around an athlete’s performance schedule. In order to set up the tissues for success, it is beneficial to treat the patient prior to a performance event. This preemptive approach is imperative when the individual has a history of specific performance-related injuries, as there will be a predisposition for potential injury. If the event involves demanding activity, such as with agility courses, it is best to do this treatment a day prior to the event. This timeframe will allow for the tissues to fully respond to the cascade of events that happen with photobiomodulation therapy. However, when dealing with a more restrained performance of a slower tempo, such as with dressage, it may be better to do this treatment just prior to the event so that we benefit from the ensuing neural blockade and endorphin release to allow for a cool, calm, collected ability to deliver.
 It is wise to provide post-event treatment as well. Even if the individual did not sustain any visible tissue injury, it makes sense to treat these sites since these structures have been worked excessively and been subjected to a higher demand of function. Inevitably, there is always some level of post-event tissue inflammation with such events, so it is important to treat after each event. A single treatment is sufficient to maintain a sound athlete, but a short course of treatments may be needed if there was an actual injury sustained during the performance. If an acute injury happens mid-event, the patient’s injuries should be treated as soon as possible.
It is wise to provide post-event treatment as well. Even if the individual did not sustain any visible tissue injury, it makes sense to treat these sites since these structures have been worked excessively and been subjected to a higher demand of function. Inevitably, there is always some level of post-event tissue inflammation with such events, so it is important to treat after each event. A single treatment is sufficient to maintain a sound athlete, but a short course of treatments may be needed if there was an actual injury sustained during the performance. If an acute injury happens mid-event, the patient’s injuries should be treated as soon as possible.
Human athletes receiving this modality have shown an ability to perform better, with increased endurance and less injury. The canine performance athlete can benefit in a similar fashion, and should be offered the benefits that photobiomodulation therapy will provide.
Welcome back to the Companion Animal Health Regenerative Medicine blog series! In today’s blog, we are going to answer three of the most frequently asked questions regarding Platelet Rich Plasma. From protocols to assessing treatment response, we will provide clarity to some of the questions surrounding this ground-breaking therapy.
Question 1: Is there any proof that Platelet Rich Plasma therapy works?
This is a very common question as there are numerous sources of information regarding the efficacy of PRP treatments. A google search will provide different answers to PRP treatments and applications, though many may not be scientifically backed or peer-reviewed (the gold standard of validating a therapy). The number one place to look for efficacy data is the pubmed.gov website. Performing a search on this website will provide the latest peer-reviewed publications which investigate efficacy (or failure) of different treatments. We have provided several of these published papers for your review, which discuss the successful treatments utilizing Platelet Rich Plasma in numerous indications and may be accessed here.
Question 2: How soon after injecting Platelet Rich Plasma do you typically see results?
It is generally accepted both in human and veterinary medicine that positive effects of this therapy will become apparent within 5-10 days post treatment.
Question 3: How do I know if the patient is responding to treatment?
There are several considerations that need to be made when trying to decipher if a patient is responding to treatment. It is important to have a method for measuring a baseline of the patient and their response to a treatment. Many practitioners using Platelet Rich Plasma therapy will have patients return to the hospital 2 weeks post-treatment and will utilize on of the following methods to measure response.
- Canine Brief Pain Survey for Pet Owners – Created by the University of Pennsylvania, this survey asks the pet owner to grade their dog’s pain and general habits which may indicate improvement from the treatment. To access this survey (for free), click here.
- Pain Assessment/Palpation – Palpation is an extremely valuable tool when assessing a dog’s pain and locating the area of injury. Palpation can not only serve as a useful diagnostic, but can also provide feedback post treatment as to if the pain is still present or has reduced from its original state.
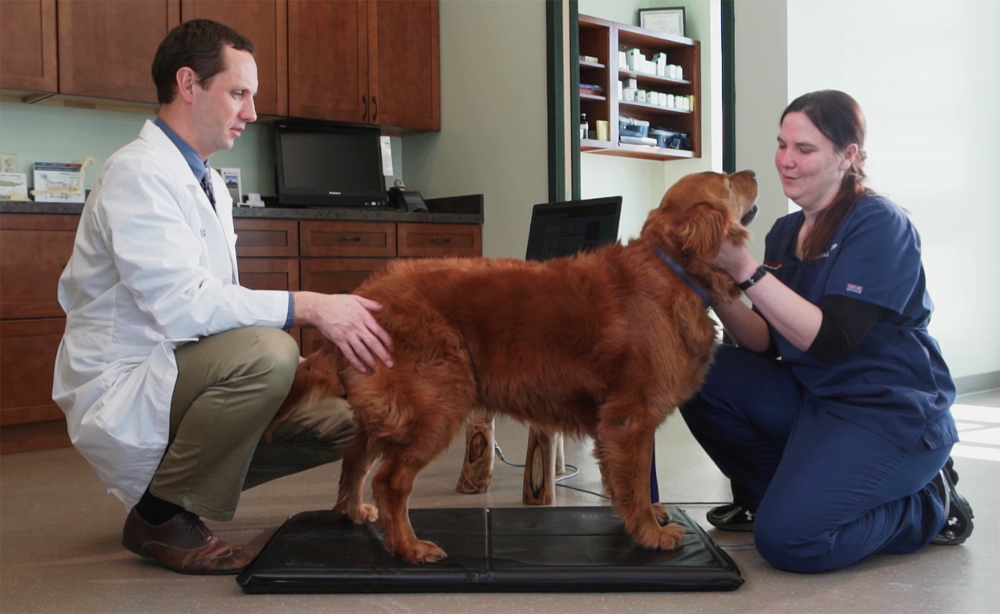
- Stance or Gait Analysis – Having a quantifiable measurement both pre-and post-treatment will provide you the best data for if a dog is improving from a therapy. Stance and Gait analysis provide objective measurements of the dog’s percent weight bearing on each of its limbs, center of gravity and stride length (gait analysis only).
- The Stance Analyzer is a cost-effective, compact system which can easily be integrated into a hospitals diagnostic program. Requiring a small foot print for floor space (approximately 2’ by 3’), the Stance Analyzer provides accurate readings of percent weight bearing, center of gravity and weight with a minimal time commitment. Once a reading is obtained, results are easy to interpret and can be saved for each patient to track responses to therapies.
- A gait analysis system typically consists of a 20-foot walkway mat which provides a detailed analysis of stride, locomotion and other advanced objective readouts. This too is a great system for quantifiable readouts, but requires additional space to perform correctly. These systems are more expensive than the Stance Analyzer and are typically utilized by universities performing research and specialty hospitals. Click here to read more about gait analysis systems.
- Gait videos – If you don’t have a stance analyzer or gait analysis but would like a visual comparison for pre- and post-treatment effects, you can use video to document your patient’s success! With the advancement of technology for smart phones, there are now applications and settings on the camera to take slow motion videos. These videos can enable you to point out changes in gait, stride length and head bobbing. They can also provide you with before and after videos to then market your regenerative treatments to your clients.
Stay tuned for our next blog where we will answer additional questions focused on Platelet Rich Plasma. If you have a question you would like answered, please contact Companion Animal Health at info@companiontherapies.com.





